Structure of Amino Acid, Peptide Bonds & Polypeptides
By: HWC
Date Uploaded: 03/06/2020
Tags: homeworkclinic.com Homework Clinic HWC Structure of Amino Acid Peptide Bonds Polypeptides amine' proteins alanine valine ribosome
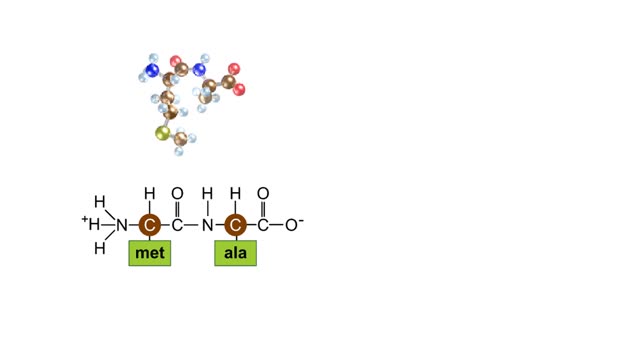
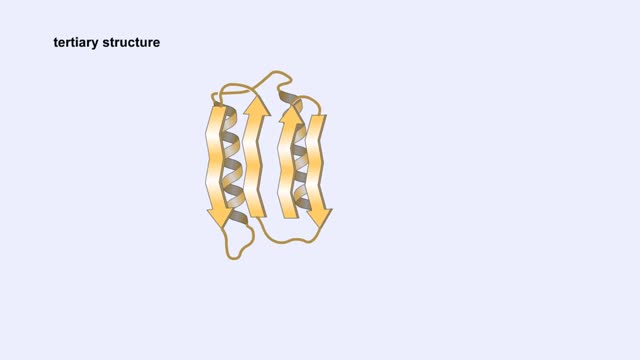
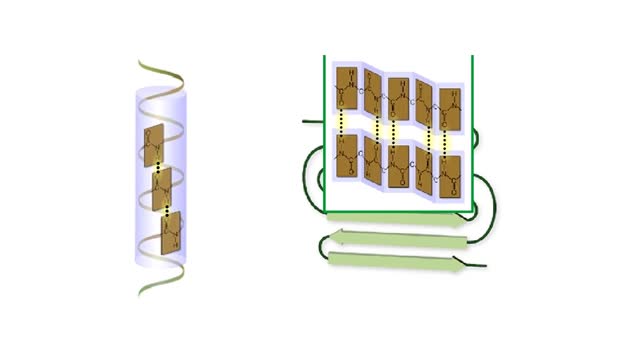
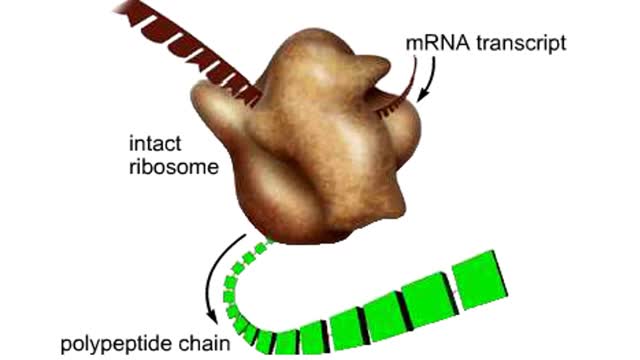
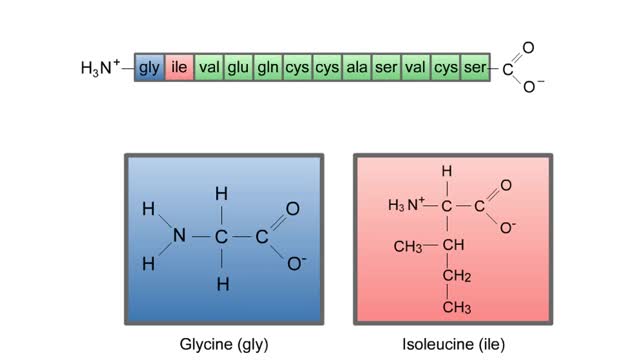
Here are the molecular formulas of three different amino acids. All amino acids share this backbone. The main difference between every amino acid is the side groups seen here, and these side groups give each of the amino acids their different characteristics. But before we get into that, let's take a closer look at the structure of an individual amino acid. You've already discovered that all amino acids share this backbone. But if you look closely, you'll see that there are differences within the backbone itself. All amino acids have a carbon atom at the center. This is the carbon atom at the center of the amino acid. This atom is essentially what holds the amino acid together as the other groups bind to it. The central carbon atom is also the point at which the other parts of the amino acid rotate. We'll learn more about how this happens and why it is important a little later. Now let's look at the amino group. An amino group is composed of nitrogen and hydrogen. This is the amino group of the amino acid. Notice that it has a nitrogen atom. The word `amine' is a Latin word that refers to any molecule that is similar to ammonia, such as this amino group. Now let's look at the carboxyl group, which is the acid part of the amino acid. A carboxyl group contains carbon, oxygen, and hydrogen. This is the carboxyl group of the amino acid. Notice that it has a carbon bound to two oxygen atoms. This is where the group gets its name: carb-oxyl. The hydrogen attached to one of the oxygen atoms is what gives the amino acid its acid properties. The final two groups are easy. The first is a hydrogen atom that is bound to the central carbon atom. The second is the side group which we discussed earlier. The side group is different for each amino acid and it will affect how the amino acid acts when it binds to other amino acids. So let's take a look at how amino acids bind to each other and make proteins. The bond between two amino acids is called a peptide bond. Now let's look at how these bonds are formed. When a peptide bond forms, a molecule of water is produced. Which atoms in these two amino acids do you think might be lost to form this water molecule? Watch this peptide bond form between alanine and valine. The carbon atom in the carboxyl group of alanine reacts with the nitrogen atom in the amino group of valine. At the same time, the alanine loses an OH group, and the valine loses a hydrogen atom, forming the water molecule. This kind of bond is repeated within a protein thousands of times using many different amino acids. In a living cell, these bonds are created in a set order for each protein on a cellular structure called a ribosome. But that will be discussed in detail in a later animation. Right now, let's take a closer look at the central carbon atom and how it affects the whole protein molecule. The central carbon atom is the point at which the other parts of the amino acid rotate. Small rotations of the bonds around the central carbon atom can lead to large changes in the overall protein structure. When you have a protein with thousands of amino acids, these small rotations can add up to a large variety of complex structures.
Add To
You must login to add videos to your playlists.
Advertisement




Comments
0 Comments total
Sign In to post comments.
No comments have been posted for this video yet.