Search Results
Results for: 'sugar molecule'
ETC Protein Complexes & Chemiosmosis (Total ATP Production and ATP Synthase)
By: HWC, Views: 10583
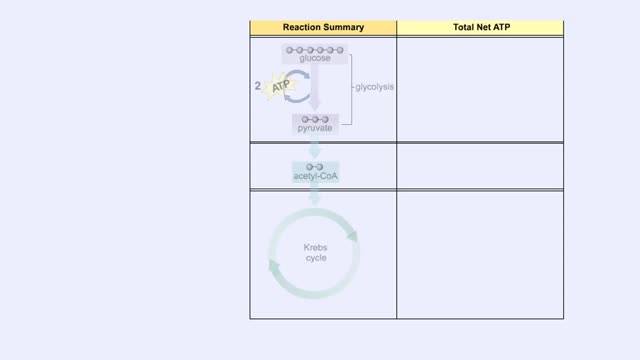
You will notice that FADH2 donates two electrons further downstream than NADH. This results in only two protons being pumped across the inner membrane. The final electron acceptor for these transported electrons is oxygen. Oxygen receives these electrons, plus protons from the aqueous matrix. ...
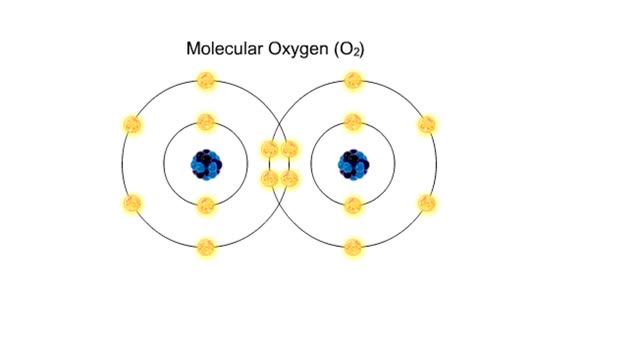
Major Elements in Biological Molecules: Carbohydrates
By: HWC, Views: 10455
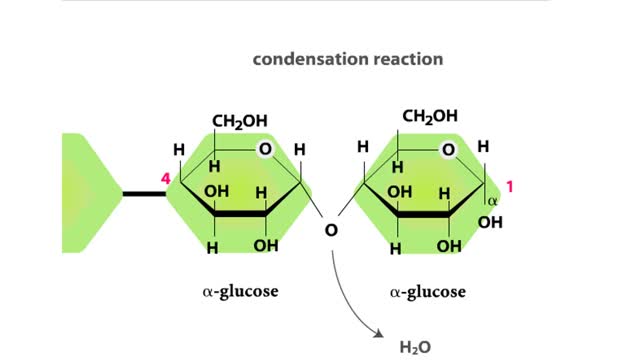
Carbohydrates include simple sugars (monosaccharides) as well as large polymers (polysaccharides). Glucose is a hexose, a sugar composed of six carbon atoms, usually found in ring form. A starch macromolecule is a polysaccharide composed of thousands of glucose units. Glucose molecules can be ...
What are Strong & Weak Acids and How they're different?
By: HWC, Views: 9667
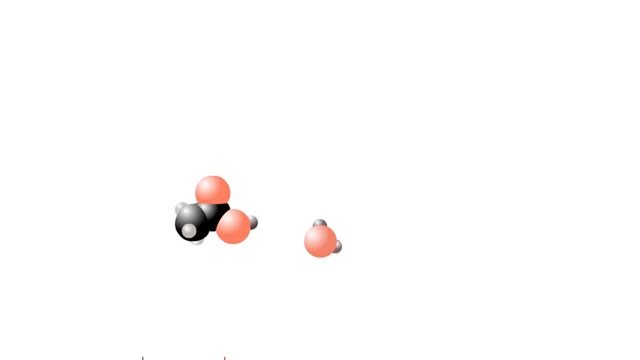
Let's consider the changes that take place when hydrogen chloride, HCI, is added to water. You will need to recognize space-filling models of HCI molecules, hydronium ions (H30+), chloride ions (C11, and water molecules (H20). They are shown at the right. When HC1 molecules dissolve in water, ...
By: HWC, Views: 9667
The slight positive charge of a hydrogen atom in a water molecule can attract an atom with a slight negative charge, such as the nitrogen in a molecule of ammonia. This forms a hydrogen bond between the two atoms. Hydrogen bonds join the two strands of a DNA molecule. Although hydrogen bo...
Bond in biological molecules (Ionic, Covalent and Hydrogen bonds)/ How atoms bond?
By: HWC, Views: 8214
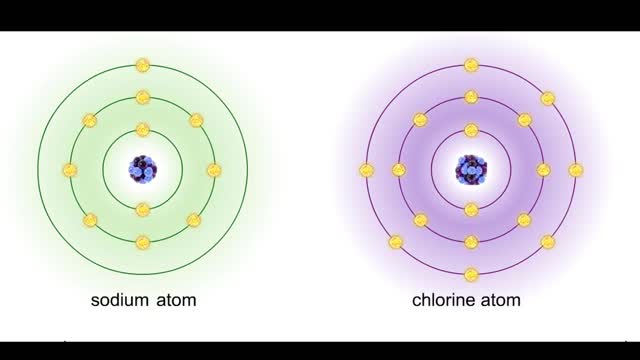
Sodium atoms and chloride atoms have unfilled orbitals in their outer shells. The lone electron in the outermost shell of a sodium atom can be pulled or knocked out. This ionizes the atom. It is now a positively charged sodium ion. A chlorine atom has an electron vacancy in its outer shell and...
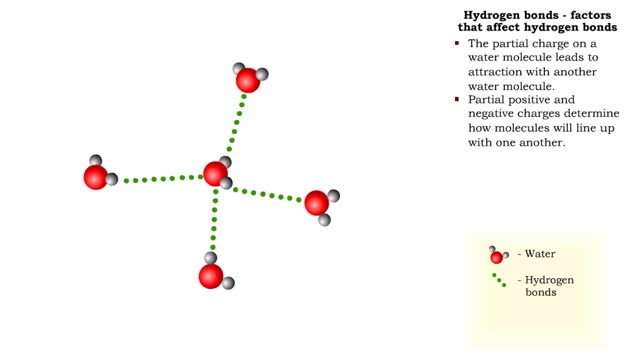
Hydrogen bonds - role in the body
By: HWC, Views: 11173
A hydrogen bond is the electromagnetic attraction between polar molecules in which hydrogen is bound to a larger atom, such as oxygen or nitrogen. This is not a sharing of electrons, as in a covalent bond. Instead, this is an attraction between the positive and negative poles of charged atoms. ...
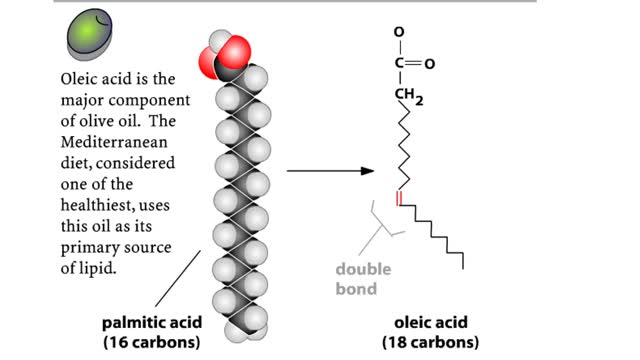
Major Elements in Biological Molecules: Lipids
By: HWC, Views: 10272
A triglyceride (also called triacylglycerol) is composed of three fatty acid molecules and one glycerol molecule. The fatty acids attach to the glycerol molecule by a covalent ester bond. The long hydrocarbon chain of each fatty acid makes the triglyceride molecule nonpolar and hydrophobic. Pa...
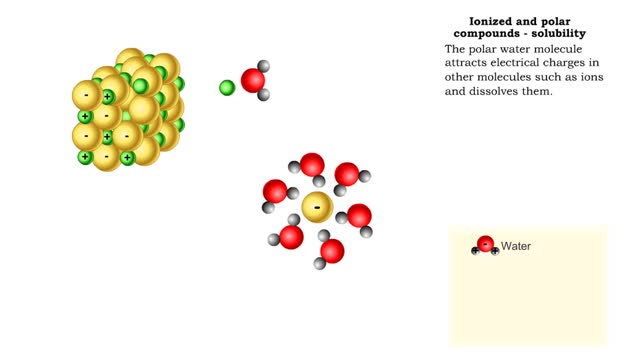
Properties of water -structure of water and polarity (Ionized and polar compounds)
By: HWC, Views: 11024
■ Water transports most of the molecules in the body. ■ The structure of a water molecule allows it to dissolve other molecules. ■ Shared electrons spend more time near the oxygen atom. ■ Oxygen end has a partial negative charge. ■ Hydrogen ends have a partial positive charge....
Sister chromatids of a metaphase chromosome animation
By: HWC, Views: 9059
At metaphase, the chromosomes are duplicated and are at their most condensed. In each chromosome. two identical sister chromatids are held together at a constricted region called the centromere. When a chromosome is condensed, interactions among chromosomal proteins keep loops of DNA tightly ...
Advertisement