Glycolysis - Introduction to ATP and the burning of sugar
By: HWC
Date Uploaded: 02/10/2020
Tags: homeworkclinic.com Homework Clinic HWC Glycolysis bunsen burner adenosine triphosphate ATP adenine base ribose sugar mitochondria chloroplasts
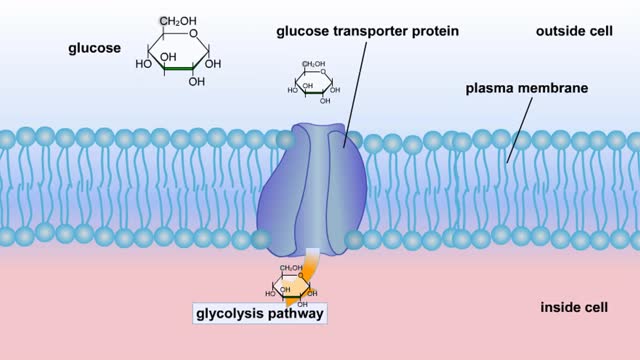
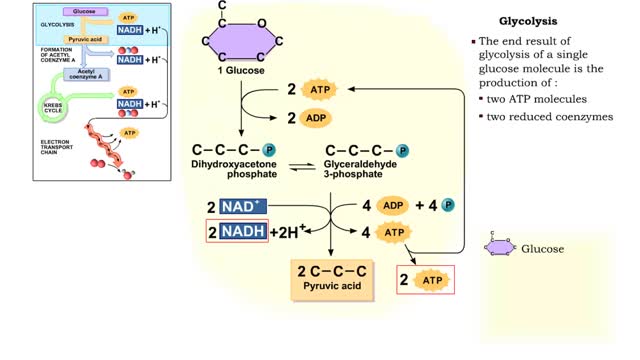
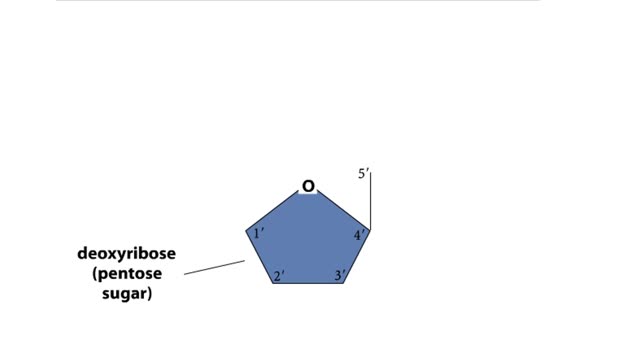
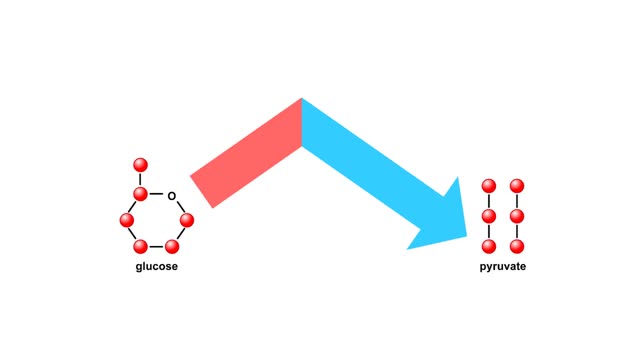
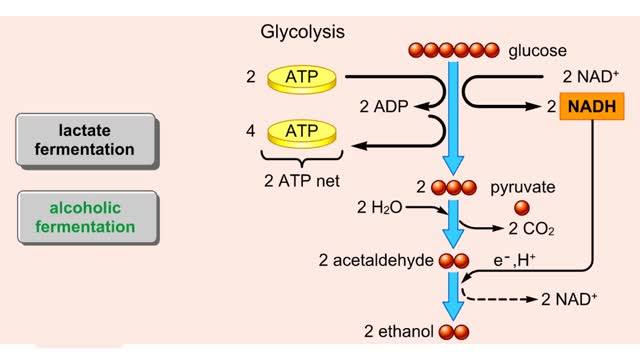
Do you use sugar with your coffee or tea? Or do you occasionally drink a sport or soft drink? As millions of people do each day, they obtain energy from the sugar added or contained in these drinks. How can we understand this concept of energy within a sugar molecule? Let's take a tablespoon of sugar and ignite it with a bunsen burner. You see that lots of energy is released during the combustion of sugar. Water and carbon dioxide are also produced. Obviously, this instant and dramatic release of energy from the combustion of sugar would kill the cell or organism. Therefore, in the cell, sugar is combusted or broken down slowly, in steps rather than instantly, with the subsequent release of useable energy in the form of an energy-carrying molecule, ATP (adenosine triphosphate). ATP or adenosine triphosphate is the principle energy-carrying molecule in the cell. It is a nucleotide that consists of three parts: an adenine base, a ribose sugar, and three phosphate groups. Energy is carried in the chemical bonds between the phosphate groups, with the terminal or third phosphate possessing the most energy. Energy is released when the terminal phosphate is cleaved. This energy can be used to fuel the many chemical reactions in the body. When ATP loses one phosphate group, ADP is formed plus inorganic phosphate. The reverse reaction which produces ATP, ADP + inorganic phosphate = ATP, takes place primarily in mitochondria and chloroplasts.
Add To
You must login to add videos to your playlists.
Advertisement



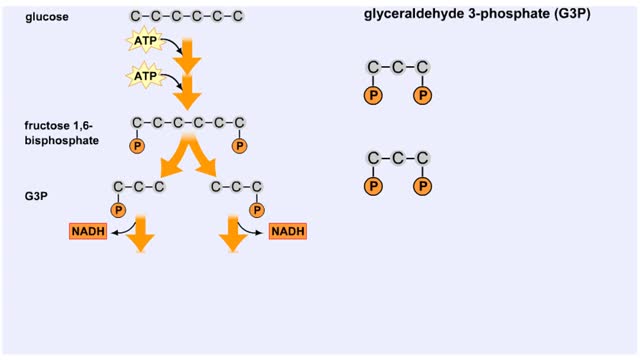
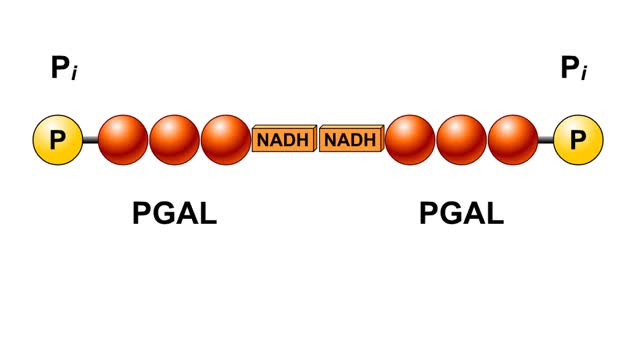
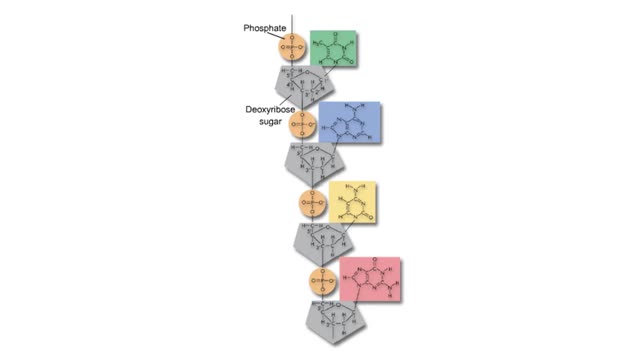
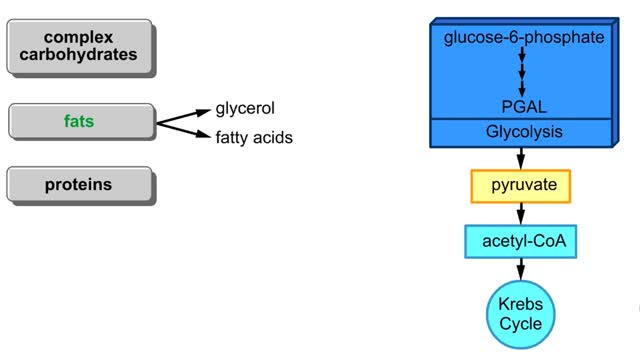
Comments
0 Comments total
Sign In to post comments.
No comments have been posted for this video yet.